
Replacing r 0 as before yields the full Kapustinskii equation. 
The derivation of the later form of the Kapustinskii equation followed similar logic, starting from the quantum chemical treatment in which the final term is 1 − d / r 0 where d is as defined above. 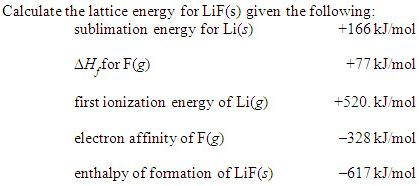
Strategy: Using Equation 8. Asked for: order of increasing lattice energy. The Born-Land equation was derived from these two following equations. Going across the periodic table, atomic radii decreases, therefore lattice energy increases. Finally, Kapustinskii noted that the Madelung constant, M, was approximately 0.88 times the number of ions in the empirical formula. Arrange GaP, BaS, CaO, and RbCl in order of increasing lattice energy. For CaF 2, calculate the lattice energy using the Born-Landé equation, the Born-Mayer equation (using d 34.5 pm), the Kapustinskii equation, and using a Born-Haber cycle.Cite your source for each datum used in these calculations. Lattice energy decreases as you go down a group (as atomic radii goes up, lattice energy goes down). The lattice energy formula can be stated as follows: LE kQ1Q2 / r. Lattice energy U L per mole may be defined as the sum of the electrostatic and repulsive energy. The total potential energy of the ionic compounds is also referred as the lattice energy. Although lattice energy can be fairly complex, Coulomb’s law is frequently used to simplify it. Lattice energy refers to the energy which is released while two oppositely charged gaseous ions attract to each other and form an ionic solid. Madelung Constant - The Madelung constant is used in determining the electrostatic potential of a single ion in a crystal by approximating the ions by point charges. The two ions attract one another, and this exothermic reaction releases energy. The strength of the bond between the ions of opposite charge in. In addition, the Born exponent, n, was assumed to have a mean value of 9. Lattice Energy - (Measured in Joule per Mole) - The Lattice Energy of a crystalline solid is a measure of the energy released when ions are combined to make a compound. Lattice Energies and the Strength of the Ionic Bond. Kapustinskii replaced r 0, the measured distance between ions, with the sum of the corresponding ionic radii. We can compute the lattice energy of nearly any ionic solid by using a modified form of Coulomb’s law. It is named after Anatoli Fedorovich Kapustinskii who published the formula in 1956. Based on the density-functional theory calculations and the established experimental data, we develop a consistent thermal EOS of platinum as a reference for pressure calibration.The Kapustinskii equation calculates the lattice energy U L for an ionic crystal, which is experimentally difficult to determine. We find that, in overall, LDA results agree best with the experiments. Special attention was paid to the influence of the electronic temperature on the phonon frequencies. Answer: UL 755 KJmol Example 2: The lattice energy of. The vibrational part was computed within the quasiharmonic approximation using density-functional perturbation theory and pseudopotentials. Lattice Energy Formula Example 1: Compute the Lattice energy of NaCl by using Born-Lande equation. 
The electronic thermal free energy was evaluated using the Mermin functional. The static lattice energy was computed by using the linearized augmented plane-wave method with local-density approximation (LDA), Perdew-Burke-Ernzerhof, and the recently proposed Wu-Cohen functional. Relationships among Ionic Lattice Energies, Molecular (Formula Unit) Volumes, and. We use density-functional theory to calculate the thermal equation of state of platinum up to 550 GPa and 5000 K. However, the established thermal equation of state (EOS) has uncertainties especially in the high P − T range. Using blackbody radiator (equal emitter and absorber of radiation at all wavelengths), Planck derived the equation for the smallest amount of energy that can be changed into light. The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Equation 4.1: U kQ1Q2 r0, where U > 0 (4.2.1) (4.2. The linear generalized equation described in this paper provides a further dimension to the prediction of lattice potential energies/enthalpies of ionic solids. Platinum is widely used as a pressure calibration standard. The first experiments towards Wave-Particle duality were done by German Physicist Max Planck (1858-1947).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
